INTRODUCTION
Rheumatoid arthritis (RA) is a chronic autoimmune disease characterized by persistent synovitis, pain, and progressive functional loss, with substantial individual and societal burden [1-3]. Its etiology and pathogenesis remain only partly elucidated, reflecting complex gene–environment interactions that sustain chronic inflammation, aberrant cellular and humoral immune responses, and synovial hyperplasia [4–13]. Histopathologically, proliferative pannus infiltrated predominantly by CD4+ T-helper lymphocytes (driven by cytokines, adhesion molecules, and chemotactic factors) invades cartilage and bone, accelerating structural damage [14–16]. Serologically, anti–cyclic citrullinated peptide antibodies (anti-CCP) complement or surpass rheumatoid factor for classification and early recognition, including seronegative presentations [17–20]. Early diagnosis remains challenging because clinical signs may be subtle and highly specific biomarkers are lacking; therefore, longitudinal assessment is essential and combines patient-reported measures (e.g., pain VAS), joint counts, acute-phase reactants (CRP, ESR), composite indices such as DAS28, and sensitive imaging (radiographs for erosions/progression; ultrasound/MRI for subclinical synovitis) [21–26].
Contemporary management follows a treat-to-target approach aiming for remission or low disease activity with csDMARDs (methotrexate as cornerstone), tsDMARDs, and bDMARDs, integrated with rehabilitation to preserve function and quality of life [27–29]. However, access to supervised exercise/education and sustained adherence outside clinic settings are uneven. Telerehabilitation—remote, structured delivery of exercise, education, and self-management with synchronous or asynchronous supervision—addresses these gaps by maintaining therapeutic “dose,” monitoring, and self-efficacy between visits. Reviews of telemedicine in RA suggest that remote care can maintain disease control and satisfaction, while underscoring the need for standardized protocols [30, 31]. Randomized trials report clinically meaningful benefits of digital models: a smartphone-supported pathway increased the proportion achieving controlled disease activity (e.g., DAS28-CRP ≤3.2) [32]; an app-based hand program improved function, pain, and work performance versus usual care [33]; and a remotely supervised weight-loss plus exercise intervention improved cardiometabolic risk, disease activity, and patient-reported outcomes in older adults with RA [34]. At RMD level, tele-delivered rehabilitation yields improvements in function and pain comparable to in-person formats in selected contexts and enables tight-control strategies with frequent assessments [35, 36].
Study aim: To compare the effectiveness of a 12-week supervised kinesitherapy program delivered as center-based rehabilitation (CR), telerehabilitation (TRG), or a hybrid combined model (CRG), versus usual care (CG), on pain, functional status, and joint tenderness in adults with RA, with outcomes assessed up to 24 weeks.
Primary objectives: To determine whether supervised rehabilitation (CR, TRG, CRG) yields greater improvements in functional status (HAQ-DI) and pain intensity (VASp) at 12 weeks compared with usual care (CG); To compare the three active pathways (CR vs. TRG vs. CRG) for superiority at 12 weeks.
Key secondary objectives: To assess sustainability of effects at 24 weeks for HAQ-DI and VASp; To evaluate changes in Tender Joint Count (TJC) across T1–T5, with between-group differences at 12 and 24 weeks; To explore adherence, safety, and tolerability across delivery modes.
MATERIALS AND METHODS
1. Study Design
We conducted a prospective cohort study over 24 months (May 2022–February 2024) in hospitals affiliated with the University of Medicine and Pharmacy Craiova, Romania. Adults with moderate rheumatoid arthritis (RA) were followed for 6 months, with standardized assessments at baseline (T1) and at 2 weeks (T2), 6 weeks (T3), 12 weeks (T4), and 24 weeks (T5). Each visit included predefined clinical and functional measures to evaluate therapeutic effectiveness.
A total of 100 patients were enrolled and allocated to four parallel groups according to the rehabilitation strategy:
• Control group (CG; n = 25) – usual pharmacologic care only;
• Telerehabilitation group (TRG; n = 25);
• Conventional (in-person) rehabilitation group (CR; n = 25);
• Combined rehabilitation group (CRG; n = 25).
Baseline characteristics were comparable across groups, including sex, age, occupation, residential background, educational level, and disease severity.
Participant retention was acceptable, with fewer than 20% discontinuing prematurely because of insufficient therapeutic response, adverse events, or withdrawal. This attrition rate supports the internal validity and reliability of the longitudinal outcome comparisons.
Baseline characteristics were statistically comparable across groups, with the only systematic difference being the rehabilitation strategy received. Evaluation of treatment effectiveness based on longitudinal change in disease activity (difference between T1 and T5) demonstrated a statistically significant effect (p < 0.05) on multiple domains: clinical indices (pain, tender and swollen joint counts), functional/quality-of-life outcomes (HAQ), and the physician/patient global assessment.
The study achieved an acceptable retention rate, with <20% of participants discontinuing prematurely (ineffective response, adverse events, or loss to follow-up). This level of attrition supports the validity and reliability of the outcomes.
2. Participants
Eligible participants were adults with rheumatoid arthritis (RA) fulfilling the 2010 ACR/EULAR classification criteria [1]. Screening followed a predefined protocol and was approved by the institutional ethics committee; all participants provided written informed consent.
Inclusion criteria
• Age ≥ 18 years;
• Moderate or higher disease activity (DAS28 ≥ 3.2), irrespective of disease duration;
• Stable antirheumatic medication for ≥ 4 weeks before baseline (no initiation or dose change of csDMARDs/tsDMARDs; no prior bDMARD exposure and no current indication to start a bDMARD);
• Glucocorticoids allowed at ≤ 10 mg/day prednisone equivalent, with no dose change within 4 weeks before baseline;
• Stable NSAID regimen for ≥ 1 month;
• Medical clearance for moderate-intensity exercise;
• Sufficient language proficiency to understand study procedures and instructions;
• Availability and willingness to attend scheduled assessments and rehabilitation sessions during follow-up.
Additional inclusion criteria for allocation to the telerehabilitation arm (TRG):
• Access to a smartphone/tablet/computer with camera and reliable internet;
• Basic digital literacy (or caregiver support) sufficient to log in, follow video sessions, and complete electronic logs;
• Home environment suitability for exercise (safe space, stable chair, non-slip surface).
Exclusion criteria
• Remission or low disease activity (DAS28 < 3.2);
• Serious comorbidities (e.g., decompensated heart failure, unstable coronary disease, uncontrolled hypertension, severe pulmonary disease), active infection, organ failure, chronic hepatitis B/C, immunodeficiency states, or active tuberculosis;
• Planned surgery or joint procedures during the 24-week follow-up;
• Intra-articular corticosteroid injection within 4 weeks prior to baseline;
• Recent initiation/escalation of csDMARDs/tsDMARDs (< 4 weeks) or oral glucocorticoid dose change (< 4 weeks);
• Conditions likely to confound RA manifestations (other inflammatory arthritides, major pain syndromes);
• Cervical spine instability/atlanto-axial subluxation or other exercise contraindications identified on screening;
• Severe psychiatric disorder, moderate-to-severe cognitive impairment, or severe visual/hearing deficits that preclude safe participation;
• Alcohol dependence;
• Lack of cooperation or inability to comply with study procedures.
This framework ensures inclusion of adults with active RA suitable for non-biologic management, standardizes medication stability to reduce confounding, and explicitly addresses exercise safety and digital readiness for the telerehabilitation arm, thereby strengthening internal validity and participant safety.
3. Study Treatments
Participants were allocated to four parallel groups receiving standardized rehabilitation pathways over 12 weeks (3 sessions/week; 36 sessions planned), with a follow-up assessment at 24 weeks.
• CG (usual care): Background pharmacologic management only. Participants received written joint-protection guidance (joint economy, activity pacing, local heat for stiffness, ergonomics for hand/wrist and spine) and were encouraged to remain physically active ad libitum without supervised kinesitherapy. Self-directed activity was recorded in logs; all scheduled study assessments were attended.
• TRG (telerehabilitation): Fully remote program, 3 session/week, delivered via a secure, GDPR-compliant video platform, supervised in real time by a licensed kinesitherapist (KT). Each session included pre-session screening (resting pain, morning stiffness, flare signs, recent medication changes) and post-session AE checks. Adherence was tracked via connection logs and home-exercise diaries.
• CR (center-based rehabilitation): In-person, hospital-based kinesitherapy 3 session/week in a rehabilitation gym (group size ≤12). Sessions were conducted under the supervision of a rehabilitation physician and at least one KT; vitals and tolerance were monitored on site.
• CRG (combined/hybrid): Hybrid pathway with six initial in-person sessions (skill acquisition, safety cues, load calibration), followed by remote supervised sessions for the remaining program (30 sessions), ensuring continuity with the same KT across settings.
Intervention content (active arms): Two integrated components: postural hygiene and aerobic/conditioning, embedded in a RA-specific session template:
• Warm-up (5–10 min; gentle whole-body mobility; cervical/thoracic/lumbar ROM in pain-free range);
• Postural hygiene and mobility (10–15 min; neutral spine cues; scapular setting; hip/knee/ankle alignment; hand/wrist ergonomics; joint-protection strategies for ADLs);
• Strength/endurance at low load (15–20 min; isometrics and low-load isotonic for major muscle groups; proximal stabilizers of shoulder/hip; closed-chain lower limb; hand function: gentle grip/pinch, tendon glides, ulnar-drift-protective patterns);
• Aerobic block (15–25 min; walking/cycling/step or arm-ergometer as available);
• Breathing/relaxation (5–10 min; diaphragmatic breathing, flexibility, relaxation).
Progression and intensity: Weeks 1–2 ≈10–20 min/session, advancing to ~60 min by weeks 4–6 and maintained thereafter; target 40–60% HR-reserve. During flares, load was reduced and re-escalated conservatively per protocol.
Safety and fidelity: Stop-rules included VAS pain >7/10, presyncope, undue dyspnea, chest pain, or abnormal vitals; events were documented and reviewed by the study physician. Intervention fidelity was ensured through a procedural manual, therapist checklists, and periodic supervision.
Concomitant care: Background antirheumatic therapy remained stable per inclusion criteria. Rescue analgesics/NSAIDs were permitted as clinically indicated and recorded. No additional physical modalities (e.g., diathermy, TECAR, TENS, manual therapy) were permitted outside the study protocol.
Adherence: Completing ≥75% of scheduled sessions defined adherence. Dose received was quantified as sessions attended × minutes completed. Reasons for missed sessions and all adverse events were recorded.
4. Parameters and Measurements
All eligible participants received standardized written and verbal information on study aims and procedures and provided written informed consent. A comprehensive baseline assessment was conducted and repeated at prespecified time points (T1 = baseline; T2 = week 2; T3 = week 6; T4 = week 12; T5 = week 24), using harmonized procedures aligned with adult RA management guidelines [27],[28]. Where feasible, assessments were scheduled at similar times of day and performed by the same trained assessor, blinded to group allocation, to minimize measurement variability.
Assessment domains and instruments:
• Joint examination: Systematic evaluation of hands, wrists, elbows, shoulders, hips, knees, ankles, and feet, recording swelling, tenderness, and active range of motion. Tender and swollen joint counts were documented using the 28-joint format (TJC28/SJC28).
• Disease activity: DAS28-ESR was the primary disease-activity measure; DAS28-CRP was analyzed in sensitivity checks when applicable. Patient and physician global assessments were collected on 0–100 scales.
• Functional status: Health Assessment Questionnaire Disability Index (HAQ-DI) (validated Romanian version) to capture limitations in activities of daily living.
• Pain and stiffness: Pain intensity on a 100-mm Visual Analog Scale (VASp); morning stiffness recorded in minutes. TJC28 and SJC28 contributed to the longitudinal symptom burden profile.
5. Ethics Approval
Participant safety and ethical conduct were central to this study involving adults with RA. All individuals were informed about study aims, procedures, potential risks/benefits, and their right to withdraw at any time without penalty or impact on standard care. Written informed consent was obtained prior to any study activity. The protocol complied with the Declaration of Helsinki and Good Clinical Practice principles. Ethical approval was granted by the Ethics Committee of the University of Medicine and Pharmacy Craiova, approval no. 26/01 March 2016.
6. Statistical Analysis
The dataset was compiled in Microsoft Excel and subsequently analyzed using IBM SPSS Statistics version 26.0 (IBM Corp., Armonk, NY, USA) and Python-based analytical tools (pandas, NumPy, SciPy; available at https://www.python.org). Analyses followed a parallel-group longitudinal framework. Baseline comparability across the four arms (CRG, TRG, CR, CG) was assessed using one-way ANOVA for continuous variables (age, BMI; reported as mean ± SD) and chi-square tests for categorical variables (sex, residence, education, professional status). When assumptions of normality (Shapiro–Wilk on residuals) or homoscedasticity (Levene) were not met, Kruskal–Wallis tests replaced ANOVA; for sparse categorical data, Fisher’s exact test was used. Longitudinal outcomes were modeled with linear mixed-effects models including fixed effects for group (CRG/TRG/CR/CG), time (T1–T5) and the group×time interaction, and a random intercept for subjects to account for within-person correlation. Where residual diagnostics suggested non-normality or heteroscedasticity, we used robust (Huber–White) standard errors as sensitivity analysis. For completeness, nonparametric sensitivity checks employed rank-based mixed models.
At specific time points, between-group contrasts were obtained from estimated marginal means (EMMs) of the mixed model, with Tukey adjustment for multiple comparisons. Where appropriate, we also report absolute change (Δ) and percent change from baseline as descriptive metrics; inferential testing relied on the mixed-model framework rather than on change scores.
Effect sizes were provided as partial η² for ANOVA and Hedges’ g (pairwise EMM contrasts) with 95% confidence intervals (CIs); for mixed models, we report marginal and conditional R². For clinical interpretability, changes in HAQ-DI were compared with established MCIDs (~0.22–0.25). All tests were two-sided with α = 0.05. Missing repeated-measures data were handled under the missing-at-random assumption inherent to mixed models; no last-observation-carried-forward was used. Analyses were conducted in R or equivalent validated software.
RESULTS
1. Baseline Characteristics of Study Groups
At baseline (T1), women predominated across all groups (CR 83.0%, CRG 79.2%, TRG 82.7%, CG 81.2%), indicating a broadly similar sex distribution (χ², p > 0.05). Mean age ranged from the early to mid-40s (CR 41.0 ± 8.5 years; CRG 43.0 ± 8.9; TRG 42.1 ± 8.1; CG 41.8 ± 8.3) with no between-group difference (one-way ANOVA, p = 0.422). Urban residence was slightly more frequent than rural across groups (CR 55.0%, CRG 53.0%, TRG 55.0%, CG 54.0%; χ², p > 0.05). Educational attainment showed a similar distribution (approximately half with secondary or higher education; χ², p = 0.723). The proportion professionally active was comparable (CR 51.0%, CRG 47.7%, TRG 50.0%, CG 49.2%; χ², p > 0.05). Body mass index (BMI) was also similar between groups (CR 22.87 ± 3.66, CRG 24.10 ± 3.21, TRG 24.01 ± 3.08, CG 23.19 ± 3.28; one-way ANOVA, p = 0.196). Overall, these demographic and anthropometric data support baseline comparability of the four groups (Table 1).
| Characteristic | CR | CRG | TRG | CG | p-value / note |
|---|---|---|---|---|---|
| Female, % | 83.0 | 79.2 | 82.7 | 81.2 | Similar distribution |
| Age, years (mean ± SD) | 41.0 ± 8.5 | 43.0 ± 8.9 | 42.1 ± 8.1 | 41.8 ± 8.3 | p = 0.422 |
| Urban residence, % | 55.0 | 53.0 | 55.0 | 54.0 | χ² test, p > 0.05 |
| Education, % | ~50% secondary/higher | ~50% secondary/higher | ~50% secondary/higher | ~50% secondary/higher | χ² test, p = 0.723 |
| Professionally active, % | 51.0 | 47.7 | 50.0 | 49.2 | χ² test, p > 0.05 |
| BMI, kg/m² (mean ± SD) | 22.87 ± 3.66 | 24.10 ± 3.21 | 24.01 ± 3.08 | 23.19 ± 3.28 | p = 0.196 |
SD=standard deviation; BMI = body mass index; p= the t-test for independent samples
2. Pain Intensity: Comparative Values and Reductions
At baseline (T1), mean VASp scores were high and broadly comparable across all groups (CRG 74.13 ± 8.53; CG 68.86 ± 9.32; TRG 72.94 ± 4.91; CR 72.88 ± 7.40; p = 0.214, one-way ANOVA). Following the treatment period, pain intensity decreased significantly in all groups, with the lowest final mean recorded in the combined rehabilitation group (CRG). At study completion (T5), mean VASp values were 28.15 ± 14.30 (CRG), 31.23 ± 13.86 (TRG), 31.67 ± 15.88 (CR), and 34.14 ± 13.94 (CG), with a significant between-group difference (p = 0.038, mixed-effects model EMMs Tukey-adjusted). Absolute changes from T1 to T5 were −45.98 (CRG), −41.71 (TRG), −41.21 (CR), and −34.71 (CG), corresponding to relative decreases between 50% and 62%. These data, summarized in Table 2, indicate a clinically meaningful and statistically supported reduction in pain across all rehabilitation programs, with the combined approach (CRG) achieving the most favorable results.
| Group | VASp T1 (mean ± SD) | VASp T5 (mean ± SD) | Δ (T5–T1) |
|---|---|---|---|
| CRG | 74.13 ± 8.53 | 28.15 ± 14.30 | −45.98 |
| CG | 68.86 ± 9.32 | 34.14 ± 13.94 | −34.71 |
| TRG | 72.94 ± 4.91 | 31.23 ± 13.86 | −41.71 |
| CR | 72.88 ± 7.40 | 31.67 ± 15.88 | −41.21 |
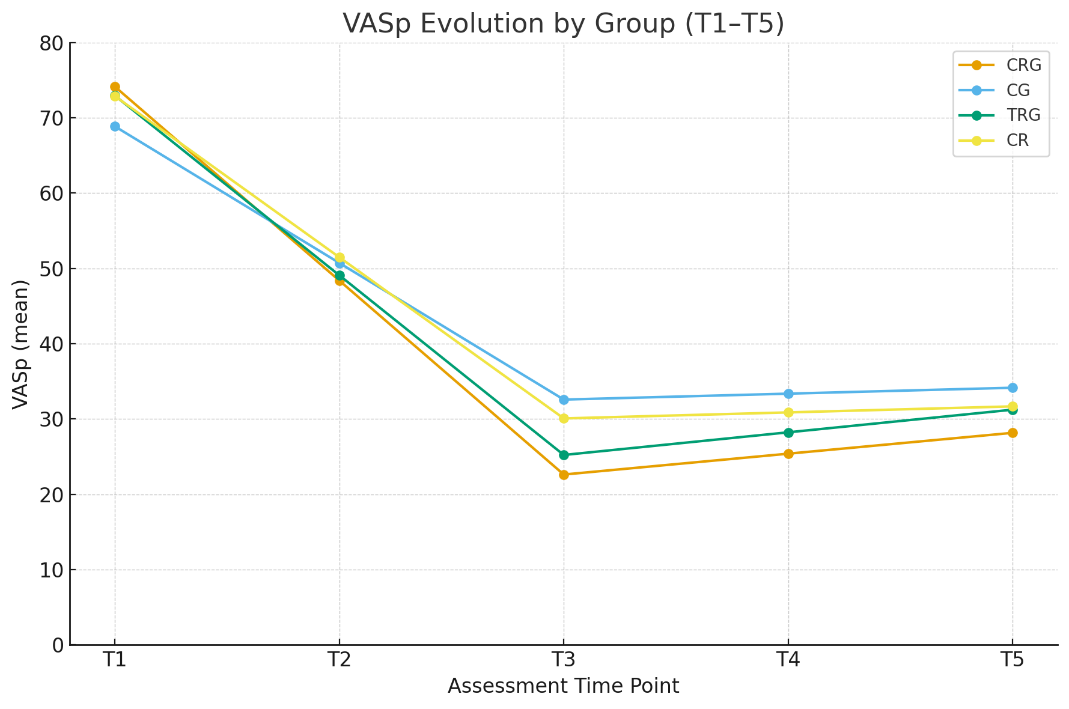
Figure 1 illustrates the evolution of VASp values across successive assessment points (T1–T5) for each study group. All rehabilitation groups (CRG, TRG, CR) showed a marked and steady decline in pain scores starting from the first follow-up, whereas the control group (CG) displayed a slower and less pronounced reduction.
The CRG curve demonstrates the steepest initial decrease and the lowest overall trajectory throughout follow-up, confirming the superior efficacy of the combined rehabilitation program in reducing pain intensity. At T3, significant between-group differences became evident (p = 0.011), and this advantage was maintained until study completion (p = 0.038).
Overall, Figure 1 visually supports the findings summarized in Table 2, showing a consistent, sustained, and clinically relevant decrease in pain for patients undergoing multimodal (combined) rehabilitation.
3. Tender Joint Count dynamics
At baseline (T1), mean TJC was comparable across groups (mean ± SD): CRG 15.46 ± 4.27, CG 16.42 ± 4.89, TRG 16.18 ± 4.79, CR 14.75 ± 3.41. Over the study period, TJC decreased in all active arms, with CRG reaching the lowest T5 value (2.30 ± 1.97). At T5, group means were: CRG 2.30 ± 1.97, TRG 2.46 ± 1.85, CR 2.93 ± 1.58, CG 4.42 ± 2.53; the between-group difference was significant (p = 0.031, estimated marginal means from a mixed-effects model, Tukey-adjusted) (Table 3).
| Group | TJC T1 (mean ± SD) | TJC T5 (mean ± SD) | Δ (T5–T1) |
|---|---|---|---|
| CRG | 15.46 ± 4.27 | 2.30 ± 1.97 | −13.16 |
| TRG | 16.18 ± 4.79 | 2.46 ± 1.85 | −13.72 |
| CR | 14.75 ± 3.41 | 2.93 ± 1.58 | −11.82 |
| CG | 16.42 ± 4.89 | 4.42 ± 2.53 | −12.00 |
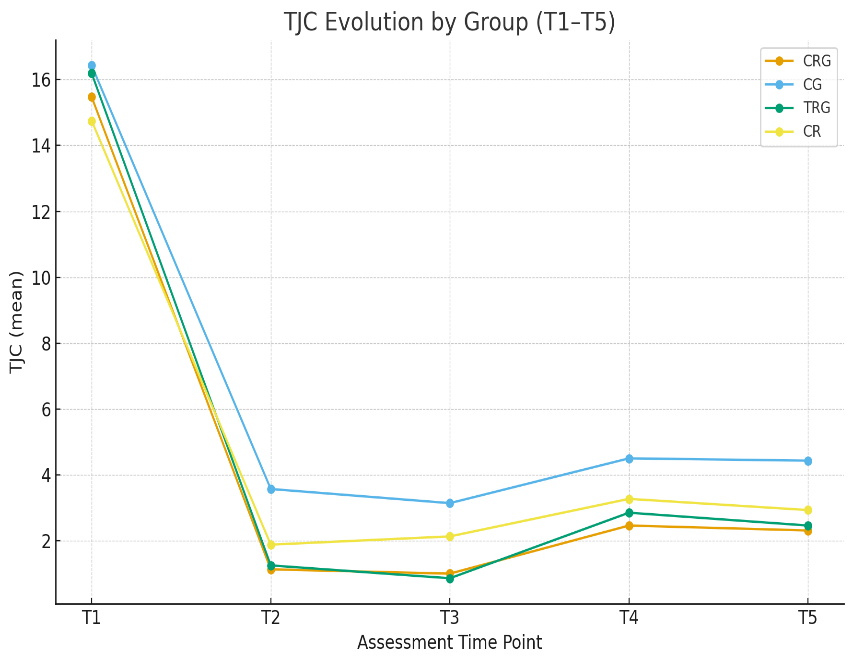
Based on these statistically significant findings, we further analyzed the dynamics of the therapeutic response reflected by the TJC across successive assessment points. In absolute terms (Figure 2), the T1→T5 reductions were TRG −13.72 (−84.8%), CRG −13.16 (−85.1%), CG −12.00 (−73.1%) and CR −11.82 (−80.1%). Between-group contrasts emerged as early as T2–T3 and generally persisted; T4 did not reach statistical significance.
Taken together, these findings support the superiority of the combined program (CRG) in achieving the lowest final TJC, while TRG yielded the largest absolute reduction, compared with in-person rehabilitation alone (CR) or usual care (CG).
4. Functional status: comparative values and changes
At baseline (T1), mean HAQ values were broadly comparable across groups (CRG 2.10 ± 0.38, TRG 2.33 ± 0.36, CR 2.21 ± 0.31, CG 1.85 ± 0.37). By T2 and T3, between-group differences were significant (T2: p = 0.0147; T3: p = 0.0036), with larger improvements in the rehabilitation arms versus control. At study completion (T5), HAQ remained below baseline in all groups, with means of 0.90 ± 0.66 (CRG), 1.00 ± 0.59 (TRG), 0.91 ± 0.46 (CR), and 1.29 ± 0.61 (CG). Absolute T1→T5 changes were −1.20 (CRG), −1.33 (TRG), −1.29 (CR), and −0.56 (CG)—all exceeding established MCIDs for HAQ (~0.22–0.25), underscoring clinical relevance. These data are summarized in Table 4
| Group (Therapy) | HAQ T1 (mean ± SD) | HAQ T5 (mean ± SD) | Δ (T5–T1) |
|---|---|---|---|
| CRG | 2.10 ± 0.38 | 0.90 ± 0.66 | −1.20 |
| CG | 1.85 ± 0.37 | 1.29 ± 0.61 | −0.56 |
| TRG | 2.33 ± 0.36 | 1.00 ± 0.59 | −1.33 |
| CR | 2.21 ± 0.31 | 0.91 ± 0.46 | −1.29 |
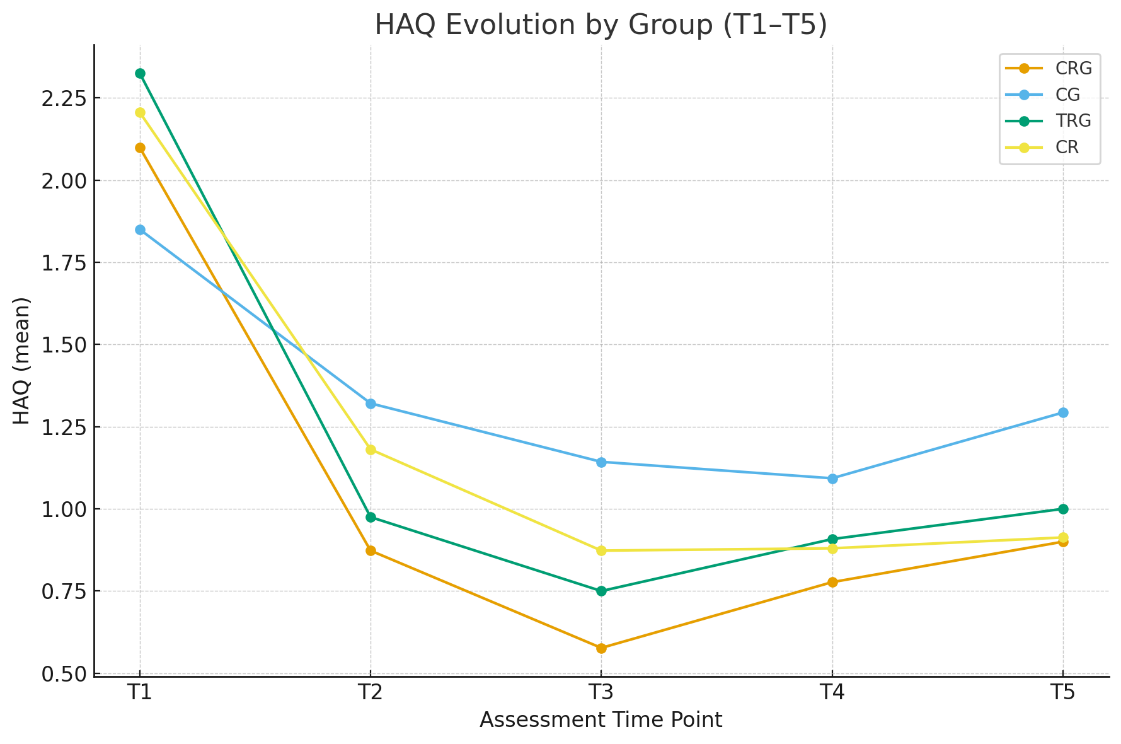
Figure 3 depicts the HAQ trajectories across T1–T5 for each group. All arms show a clear downward trend, with the combined rehabilitation group (CRG) exhibiting the steepest decline and lowest trajectory during mid-follow-up; between-group differences are significant at T2 (p = 0.0147) and T3 (p = 0.0036).
Although the contrast is not retained at T4, end-of-study values remain markedly below baseline in all groups, with CRG retaining the lowest final mean and CG the least favorable profile.
The figure 3 visually corroborates the pattern reported numerically in Table 4, indicating sustained, clinically meaningful functional improvement—most pronounced for the combined program.
DISCUSSION
In our study, pain intensity (VASp) declined markedly in all supervised arms, with the hybrid program (CRG) achieving the lowest mid-term and favorable end-of-study values versus single-modality delivery and usual care. The absolute T1 toT5 reductions are clinically meaningful and consistent with previous trials and meta-analyses showing that structured, progressive exercise produces small-to-moderate but robust analgesic effects in rheumatoid arthritis [37–40]. Our temporal pattern (rapid early decline through the 12-week program, sustained benefit at 24 weeks) mirrors the kinetics reported in supervised interventions where dose, progression, and feedback are maintained [38–41].
Mechanistically, the analgesic response likely reflects exercise-induced hypoalgesia through central descending inhibition (opioid, serotonergic, endocannabinoid pathways) coupled with peripheral effects (improved synovial perfusion, reduced stiffness) and psychobehavioral gains (reduced fear-avoidance, better self-efficacy) [42–44]. The small attenuation of between-group significance at T4 in our cohort is plausible given interindividual variability, minor flares, or fluctuations in adherence, phenomena also noted in longitudinal syntheses when training load is not further escalated after the intensive phase [38, 40].
Of note, telerehabilitation tracked center-based outcomes closely, whereas the hybrid pathway showed a consistent edge at key time points. This aligns with reports that well-designed, supervised tele-delivery can be non-inferior to in-person care for pain and function in RA and related musculoskeletal conditions (provided there is real-time supervision, individualized progression, and adherence monitoring) [41, 45, 46]. Pragmatically, the hybrid approach may leverage front-loaded in-person skill acquisition (movement quality, load calibration, reassurance) while preserving dose and supervision remotely, thereby sustaining the analgesic response with fewer logistical barriers.
Overall, our VASp findings converge with the wider literature: exercise is an effective adjunct to pharmacotherapy for pain control in RA, and delivery mode per se is less critical than program quality (progression, supervision, fidelity). When those elements are secured, tele- and hybrid models can deliver analgesic benefits comparable to, or slightly better than, traditional center-based care—particularly in terms of maintenance at follow-up [41, 45, 46].
The progressive decline in TJC observed across all active rehabilitation arms confirms that structured and supervised kinesitherapy contributes to the reduction of inflammatory activity and joint tenderness in RA. This finding aligns with earlier studies showing that individualized exercise programs can decrease disease activity scores and improve joint function when delivered under adequate supervision and intensity control [47–49]. The present results revealed statistically significant improvements in TJC as early as T2–T3, followed by a transient loss of significance at T4, and reappearance of between-group differences at T5. A similar pattern was described by Häkkinen et al., who reported early reductions in disease activity and pain perception after a 12-week strength and aerobic training program, with slight variations in the mid-term follow-up [50]. Likewise, Stenström et al. demonstrated that moderate-intensity exercise leads to consistent improvement in tender and swollen joint counts, sustained over 24 weeks [51]. The reemergence of statistical significance at the end of the study (T5) supports the sustained anti-inflammatory and analgesic effects of the hybrid program, possibly through better adherence and maintenance of neuromuscular control. Regular low-load movement promotes synovial perfusion and modulates pain transmission via endogenous opioid and serotonergic pathways [43, 52], mechanisms that may explain the consistent reduction of TJC in our participants. Overall, these findings suggest that combined rehabilitation (CRG), which integrates supervised face-to-face sessions with continued remote monitoring, may be more effective in stabilizing the clinical improvement of joint tenderness over time, compared with monomodal strategies.
In our cohort, HAQ improved substantially and early across the supervised arms. The T1 toT5 mean changes) are not only statistically significant but also well above the established HAQ MCID (~0.22–0.25), indicating clinically meaningful gains in daily function [53, 54]. The temporal pattern—rapid improvement during the 12-week program (culminating at T3), followed by maintenance at 24 weeks, is consistent with trials and meta-analyses showing that structured aerobic and strengthening programs produce moderate-to-large functional benefits in RA, particularly when dose and progression are supervised [38, 55–57]. Our finding that combined (hybrid) supervision retained the lowest final mean HAQ, with telerehabilitation closely tracking center-based delivery, aligns with reports that well-designed, supervised exercise—irrespective of contact modality—improves disability, and that remote delivery can be non-inferior when progression, feedback, and adherence monitoring are ensured [41, 45, 56, 57]. Mechanistically, improvements in HAQ likely reflect multifactorial gains: pain modulation and kinesiophobia reduction, proximal stabilization and motor control, and aerobic reconditioning that supports ADL performance [38, 42, 57]. The loss of significance at T4 in our data plausibly reflects transient variability (minor flares, attendance fluctuation), a phenomenon also noted in longitudinal syntheses where effect sizes can plateau or oscillate after the intensive phase if training dose is not escalated [55, 56]. Nevertheless, the T5 maintenance suggests that the program achieved a durable functional trajectory, a feature repeatedly observed when supervision and progression are preserved and home-based practice is supported [41, 42, 45, 56]. Taken together, our HAQ outcomes are concordant with the broader evidence base: supervised, progressive exercise is an effective adjunct to pharmacologic care in RA; hybrid models may offer a small but pragmatic advantage by combining early skill acquisition in-person with tele-supervised continuity, helping sustain functional gains beyond the supervised phase [41, 45, 56, 57].
Several limitations should be acknowledged. First, the sample size, while adequate for detecting within-group effects, may have limited the power of between-group comparisons at certain time points. Second, blinding of assessors could not be fully guaranteed due to the nature of the rehabilitation interventions, introducing potential observation bias. Third, although background pharmacologic therapy was kept stable, minor individual variations in medication adherence or analgesic use could have influenced pain perception and joint scores. Fourth, objective activity monitoring was not implemented, and home adherence relied partly on self-reported logs, which may overestimate compliance. Finally, the study was conducted at a single center with motivated participants, which may restrict external generalizability. Despite these constraints, the consistent and clinically meaningful improvements across all rehabilitation pathways support the internal validity and practical relevance of the findings.
CONCLUSIONS
This study demonstrates that structured and supervised kinesitherapy significantly improves pain, functional capacity, and joint tenderness in patients with rheumatoid arthritis. Among the three delivery modes, the hybrid (combined) model achieved the most favorable and durable outcomes, while both center-based and tele-supervised programs remained clearly superior to usual care. These findings highlight that, when content, intensity, and supervision are standardized, rehabilitation can be effectively implemented in multiple formats, including remote or hybrid settings, without loss of clinical benefit. Integrating such programs into routine RA management may increase accessibility, adherence, and long-term disease control.